Hw7 - Dr chi's class - Homework 7 Draw an energy diagram to show that kinetic and thermodynamic - StuDocu

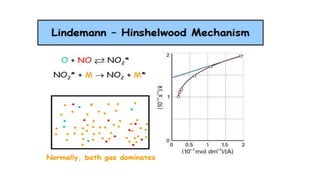
SOLVED:10 _ The Lindemann-Hinshelwood mechanism (1921) was the first to successfully explain gas phase uni-molecular reactions. For gas molecules A, the following reactions are involved. Activation: A + A +A* +A 44! =

Comparison of Exponential and Biexponential Models of the Unimolecular Decomposition Probability for the Hinshelwood–Lindemann Mechanism,The Journal of Physical Chemistry Letters - X-MOL

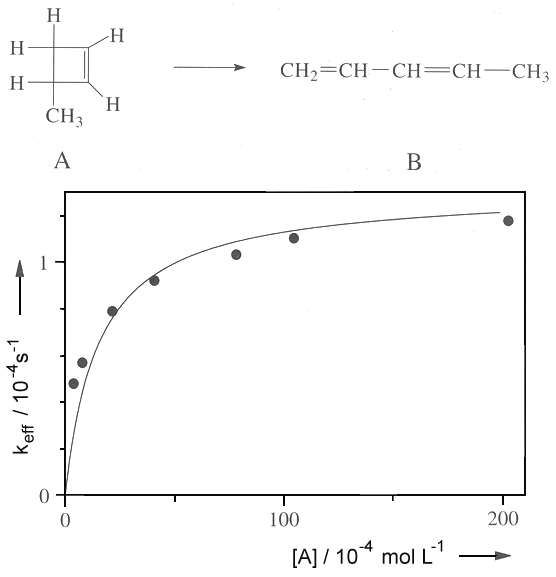
SOLVED:4) Partial pressures are frequently used instead of molar concentrations for reactions of gases that follows the ideal gas law. Consider the modified Lindemann-Hinshelwood mechanism that is employed for unimolecular reactions in